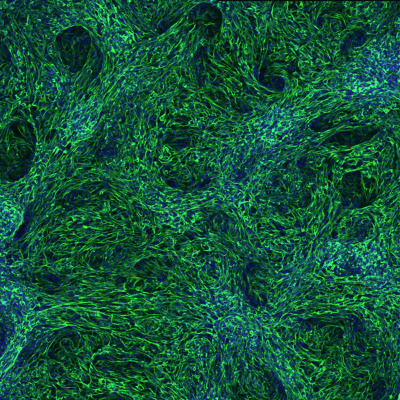
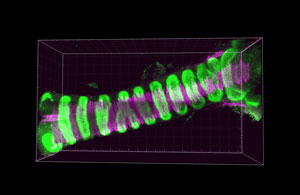
Team Director
Mitsuru Morimoto
Ph.D.
Laboratory for Lung Development and Regeneration
LocationKobe / Developmental Biology Buildings
E-mailmitsuru.morimoto@riken.jp
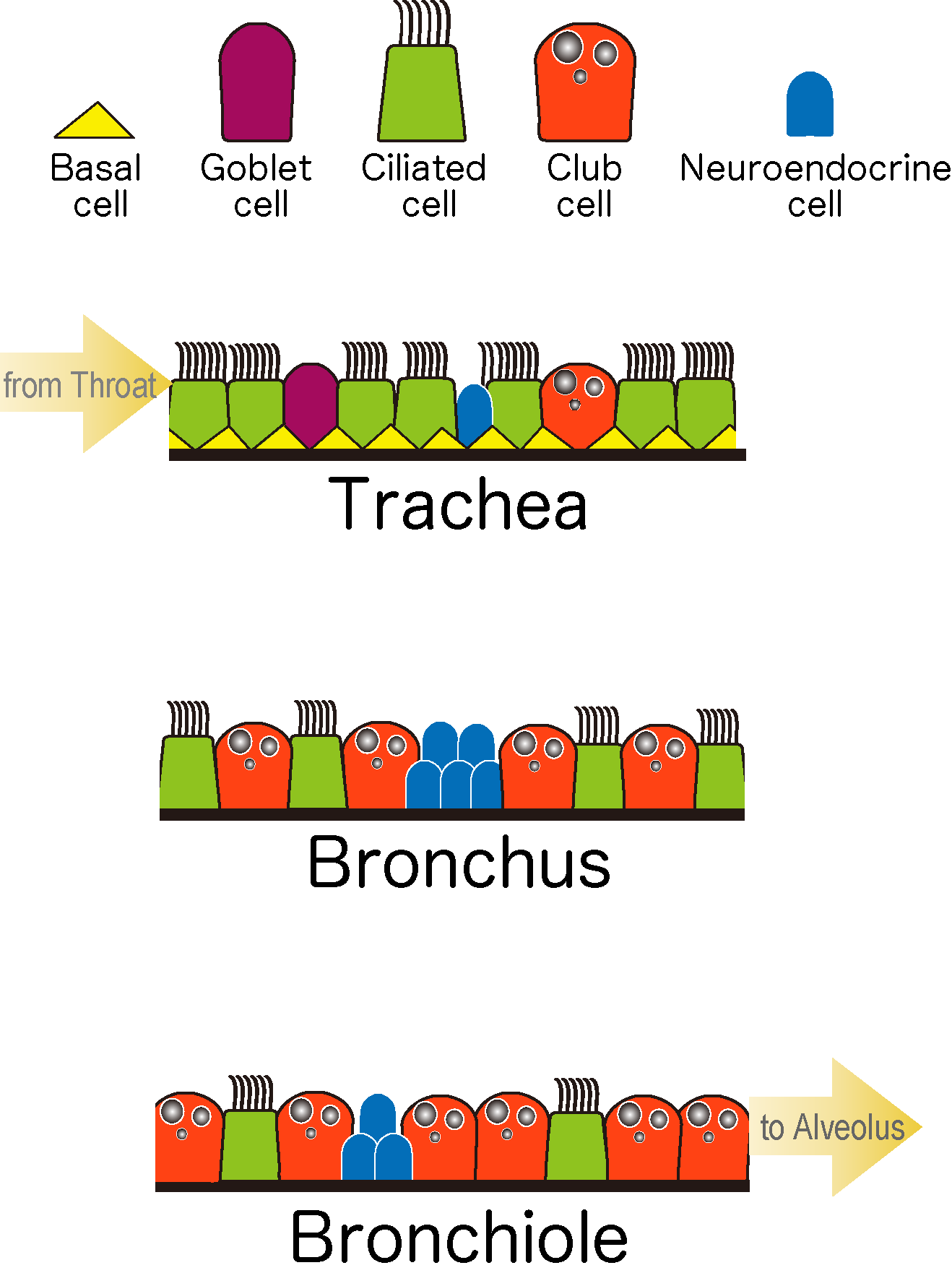
The organs of the body all play critical functional roles, which are made possible by the arrangement of differentiated cells into the structures specific to that organ. Such structures are formed throughout development, with the late embryonic and immediate postnatal periods being particularly important for the functional maturation of organ systems. Defects that arise during these organogenetic processes are closely linked to a wide range of diseases, while after birth, the body is constantly exposed to potentially damaging environmental stresses. The adult body does manifest a certain degree of regenerative ability, although this is by no means complete. To study organ formation, repair and regeneration, we have focused our research on the respiratory system in mouse. Respiratory organs in higher mammals are characterized by their efficient gas exchange, enabled by the functions of specialized cells. The development of such organs relies on the coordinated activities of both epithelial and mesenchymal tissue types, which arise from tissue-specific populations of stem cells in the developing embryo. The epithelial tissues of the conducting airways serve as the channel for the intake and exhalation of gases in the respiratory cycle, and are composed mainly of Club, ciliated, and neuroendocrine cells. In our work to date, we have shown how these various cells interact and exchange information to maintain the appropriate balance in their respective cell numbers and distributions.
Despite its location in the body’s interior, the respiratory tract is constantly exposed to environmental factors, such as infections by viruses and bacteria, smoke, and chemical toxins that may damage the airway epithelium. This damage is rapidly repaired by regenerating epithelial cells supplied by somatic stem cells in the adult tissue, and as in development, the numbers and distribution of cells in the epithelium must be maintained at levels appropriate to each region of the airway.
We focus on issues of how tissue morphology influences the formation of stem cell/niche tissue complexes in the development, repair and regeneration of respiratory organs, as well as mechanisms regulating cell proliferation and differentiation in developmental and regenerative processes.
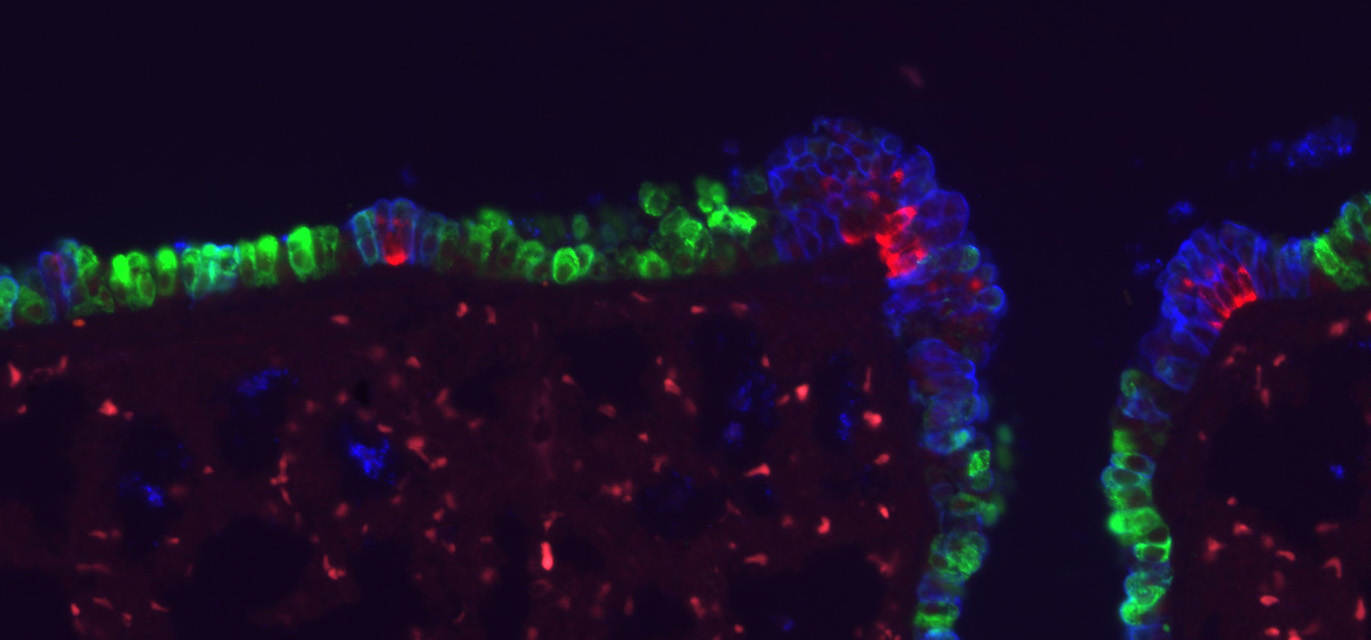
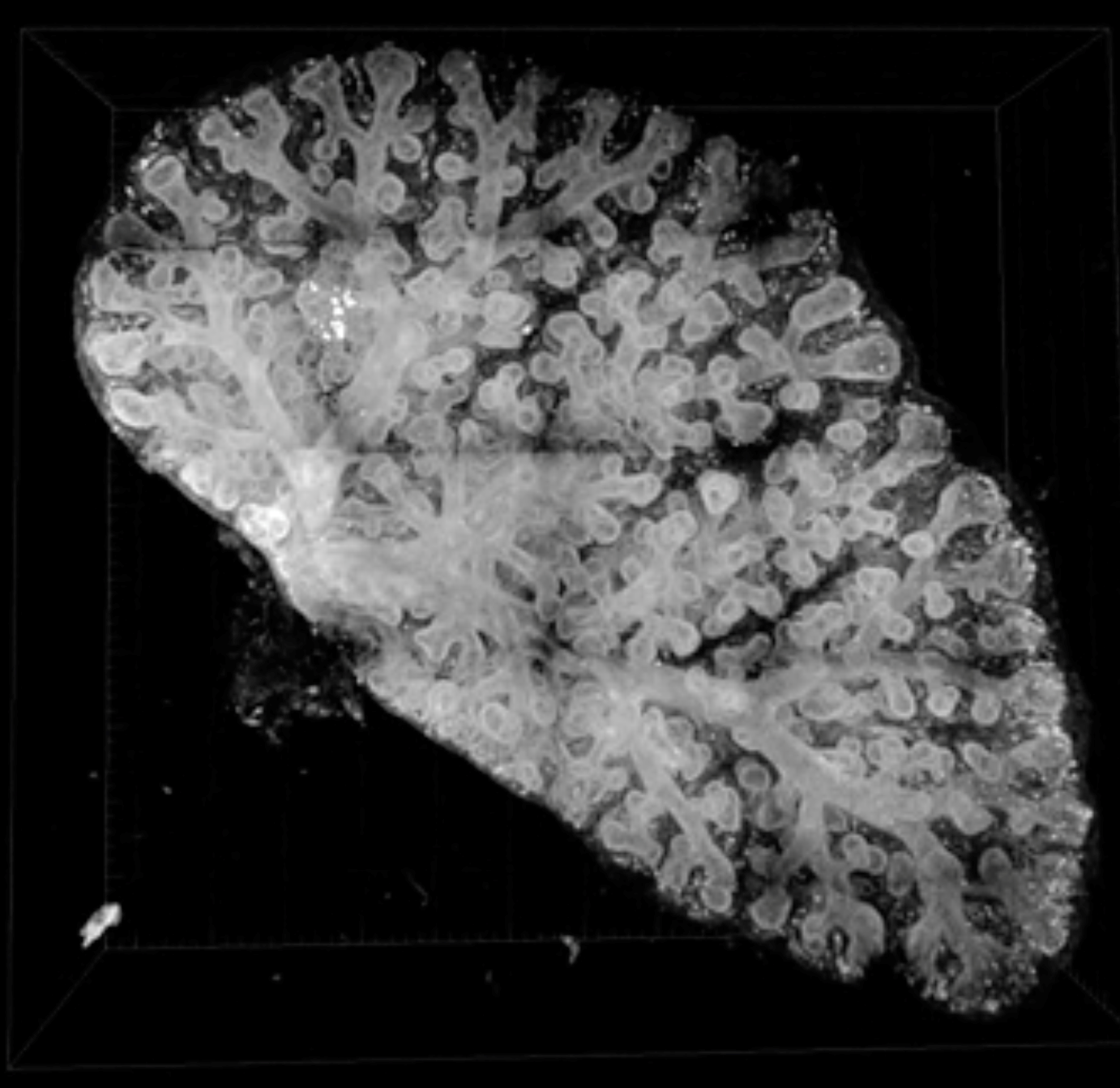
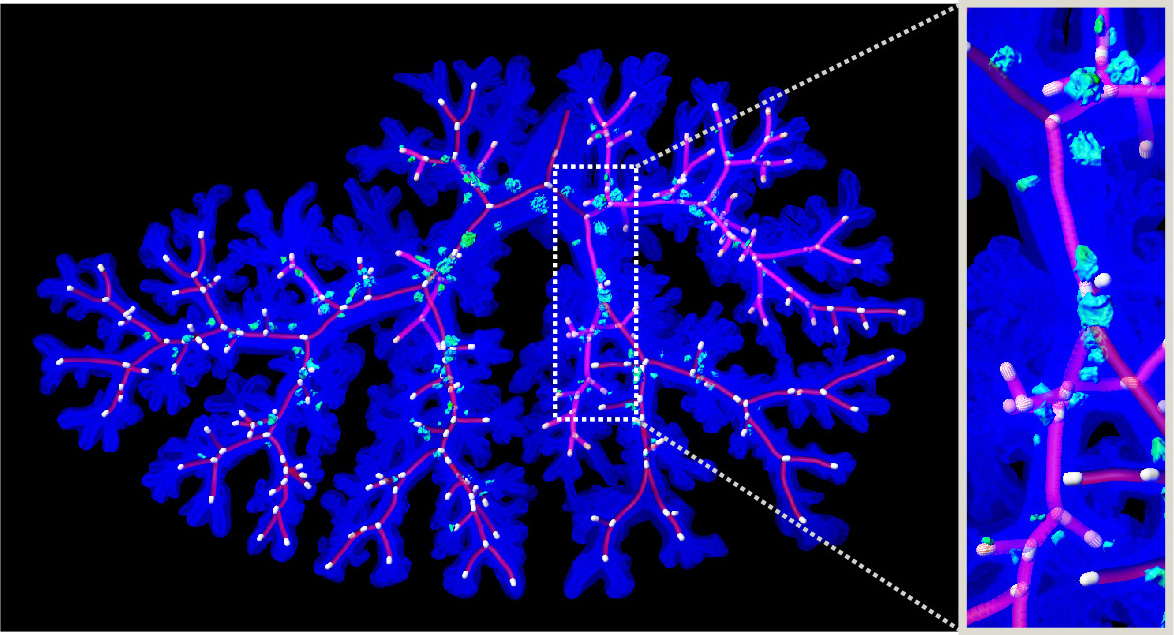
Research Theme
- Morphogenesis and regeneration biology of the lung.
- Relationship between the organ tissue structures and the stem/niche complex.
- Tubulogenesis as integrated process with individual cell shape and proliferation.
Selected Publications
Enomoto Y, Katsura H, Fujimura T, et al.
Autocrine TGF-β-positive feedback in profibrotic AT2-lineage cells plays a crucial role in non-inflammatory lung fibrogenesis.
Nature Communications
14(1), 4956 (2023)
doi: 10.1038/s41467-023-40617-y
Fujimura T, Enomoto Y, Katsura H, et al.
Identifying a Lung Stem Cell Subpopulation by Combining Single-Cell Morphometrics, Organoid Culture, and Transcriptomics.
Stem Cells
41(8), 809-820 (2023)
doi: 10.1093/stmcls/sxad044
Kishimoto K, Iwasawa K, Sorel A, et al.
Directed differentiation of human pluripotent stem cells into diverse organ-specific mesenchyme of the digestive and respiratory systems.
Nature Protocols
17(11), 2699-2719 (2022)
doi: 10.1038/s41596-022-00733-3
Kiyokawa H, Yamaoka A, Matsuoka C, et al.
Airway basal stem cells reutilize the embryonic proliferation regulator, Tgfβ-Id2 axis, for tissue regeneration.
Developmental Cell
56(13), 1917-1929 (2021)
doi: 10.1016/j.devcel.2021.05.016
Kishimoto K, Furukawa KT, Luz-Madrigal A, et al.
Bidirectional Wnt signaling between endoderm and mesoderm confers tracheal identity in mouse and human cells.
Nature Communications
11, 4159 (2020)
doi: 10.1038/s41467-020-17969-w
Kishimoto K, Tamura M, Nishita M, et al.
Synchronized mesenchymal cell polarization and differentiation shape the formation of the murine trachea and esophagus.
Nature Communications
9, 2816 (2018)
doi: 10.1038/s41467-018-05189-2
Tsao P, Matsuoka C, Wei SC, et al.
Epithelial Notch signaling regulates lung alveolar morphogenesis and airway epithelial integrity.
Proceeding of the National Academy of Sciences of the United States of America
113, 8242-8247 (2016)
doi: 10.1073/pnas.1511236113
Noguchi M, Sumiyama K, Morimoto M.
Directed migration of pulmonary neuroendocrine cells toward airway branches organizes the stereotypic location of neuroepithelial bodies.
Cell Reports
13, 2679-2686 (2015)
doi: 10.1016/j.celrep.2015.11.058
Morimoto M, Nishinakamura R, Saga Y, Kopan R.
Different assemblies of Notch receptors coordinate the distribution of the major bronchial Clara, ciliated and neuroendocrine cells.
Development
139, 4365-4373 (2012)
doi: 10.1242/dev.083840
Morimoto M, Liu Z, Cheng HT, et al.
Canonical Notch signaling in the developing lung is required for determination of arterial smooth muscle cells and selection of Clara versus ciliated cell fate.
Journal of Cell Science
123, 213-224 (2010)
doi: 10.1242/jcs.058669
Morimoto M, Kopan R.
rtTA toxicity limits the usefulness of the SP-C-rtTA transgenic mouse.
Developmental Biology
325, 171-178 (2009)
doi: 10.1016/j.ydbio.2008.10.013
Morimoto M, Takahashi Y, Endo M, Saga Y.
The Mesp2 transcription factor establishes segmental borders by suppressing Notch activity.
Nature
435, 354-359 (2005)
doi: 10.1038/nature03591