
Team Director
Wataru Kimura
Ph.D.
Laboratory for Heart Regeneration
Location Kobe / Developmental Biology Buildings
E-mail wataru.kimura[at]riken.jp
Please replace [at] with @.
Heart disease is the leading cause of death worldwide. The main reason for this is our inability to regenerate damaged myocardium in the heart. Proliferation of cardiomyocytes (heart muscle cells), is a major mediator of mammalian heart regeneration in neonates and myocardial turnover in adults. However, little is known about the mechanisms regulating the cardiomyocyte cell cycle. We have recently shown that a rapid increase in mitochondrial respiration and in oxidative stress induce cell cycle arrest in neonatal cardiomyocytes. Our research interest focuses on how the postnatal mammalian heart loses regenerative capacity following injury- and age-related myocardial damage, and whether it is possible to re-awaken endogenous regenerative capacity. We utilize molecular and cellular tools and mouse genetics to understand the role of hypoxia signaling and oxidative stress in cardiomyocyte cell cycle regulation throughout the life cycle of mammals.
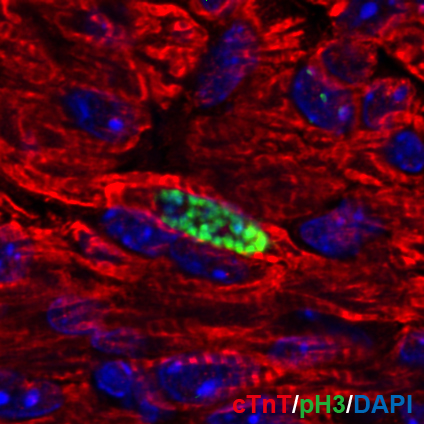
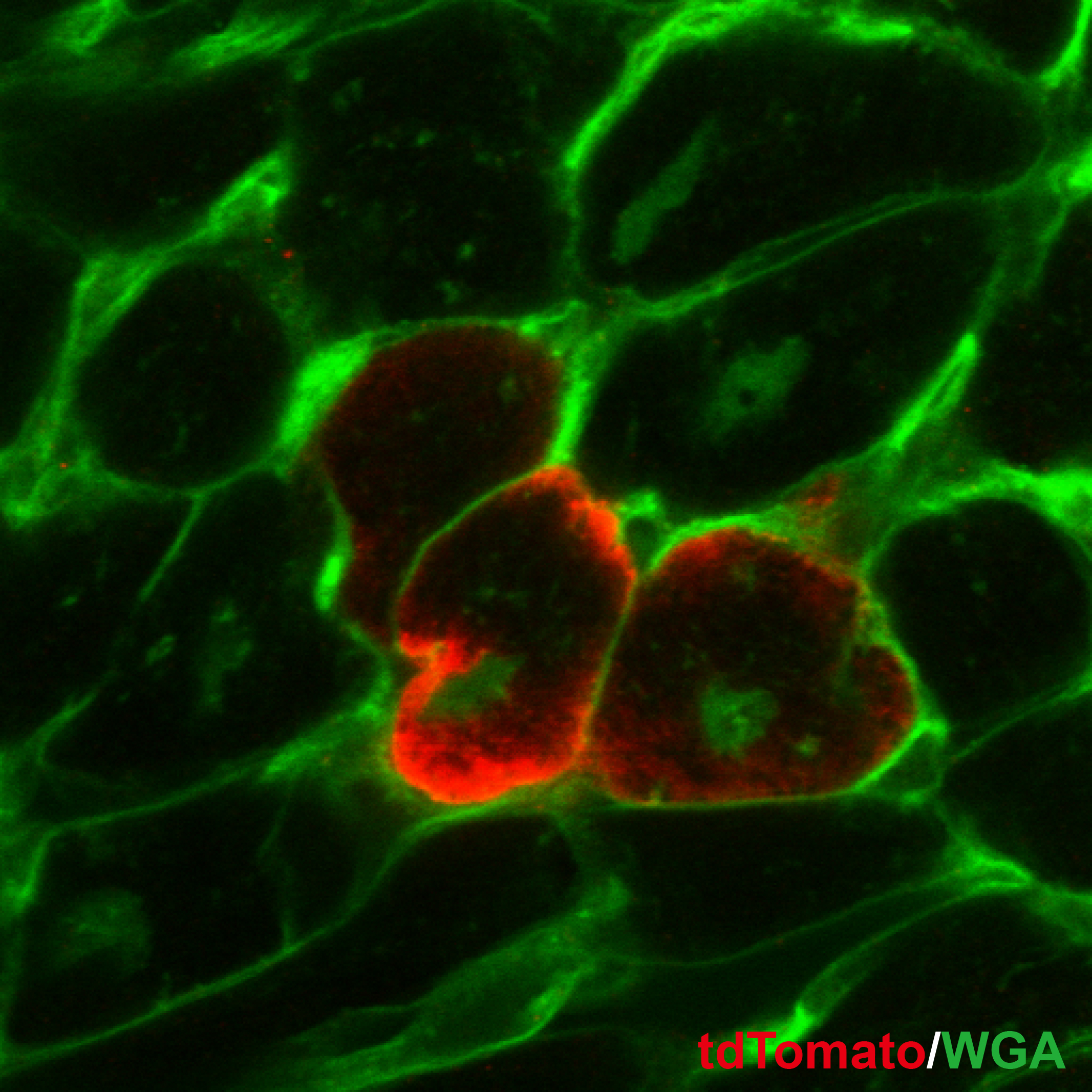
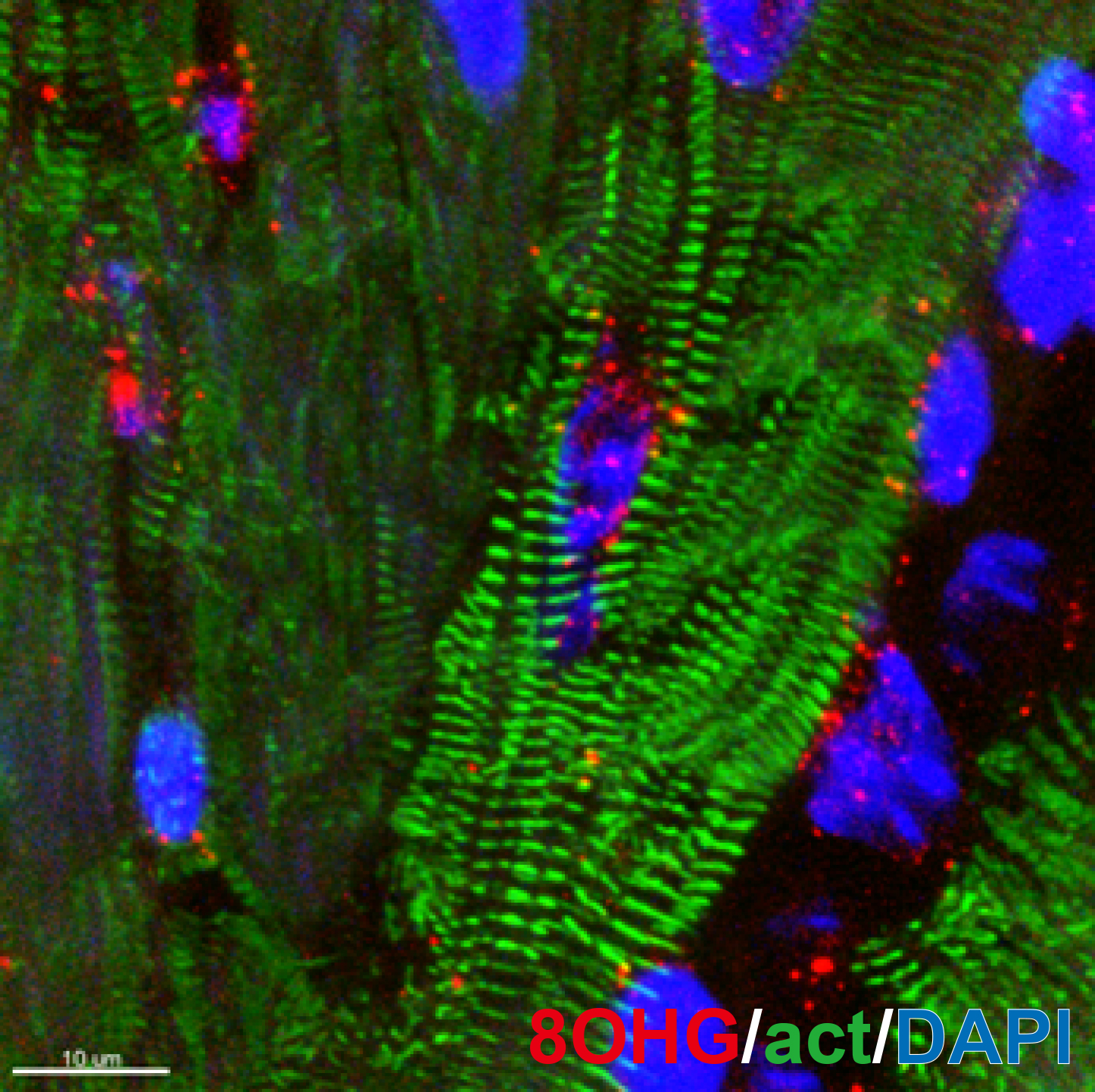
Research Theme
- Dynamic change in kinetics of postnatal cardiomyocyte renewal
- Role of hypoxia signaling in cardiomyocyte renewal
- Induction of myocardial regeneration by engineering oxidative metabolism
Selected Publications
Sakaguchi A, Kawasaki M, Saito Y, et al.
Benzyl isothiocyanate induces heart regeneration.
iScience
28, 112724 (2025)
doi: 10.1016/j.isci.2025.112724
Xiao F, Nguyen N, Wang P, et al.
Adducin Regulates Sarcomere Disassembly During Cardiomyocyte Mitosis
Circulation
150(10), (2024)
doi: 10.1161/CIRCULATIONAHA.122.059102
Saito Y, Sugiura Y, Sakaguchi A, et al.
Postnatal xanthine metabolism regulates cardiac regeneration in mammals
bioRxiv
(2024)
doi: 10.1101/2024.07.24.605040
Nishiyama C, Saito Y, Sakaguchi A, et al.
Prolonged Myocardial Regenerative Capacity in Neonatal Opossum.
Circulation
146(2), 125-139 (2022)
doi: 10.1161/CIRCULATIONAHA.121.055269
Sakaguchi A, Kawasaki M, Saito Y, et al.
Benzyl isothiocyanate induces cardiomyocyte proliferation and heart regeneration
bioRxiv
146, (2022)
doi: 10.1101/2021.09.08.459197
Sakaguchi A, Kimura W.
Metabolic regulation of cardiac regeneration: roles of hypoxia, energy homeostasis, and mitochondrial dynamics
Current Opinion in Genetics & Development
70, 54-60 (2021)
doi: 10.1016/j.gde.2021.05.009
Saito Y, Kimura W.
Roles of phase separation for cellular redox maintenance
Frontiers in Genetics
12, 691946 (2021)
doi: 10.3389/fgene.2021.691946
Sakaguchi A, Nishiyama C, Kimura W.
Cardiac Regeneration as an Environmental Adaptatio
Biochimica et Biophysica Acta - Molecular Cell Research
1867(4), 118623 (2020)
doi: 10.1016/j.bbamcr.2019.118623
Nakada Y, Canseco D C, Thet S, et al.
Hypoxia induces heart regeneration in adult mice.
Nature
541, 222-227 (2017)
doi: 10.1038/nature20173
Kimura W, Xiao F, Canseco D C, et al.
Hypoxia fate mapping identifies cycling cardiomyocytes in the adult heart.
Nature
523, 226-230 (2015)
doi: 10.1038/nature14582
Canseco D C, Kimura W, Garg S, et al.
Human ventricular unloading induces cardiomyocyte proliferation.
Journal of the American College of Cardiology
65, 892-900 (2015)
doi: 10.1016/j.jacc.2014.12.027
Puente B N, Kimura W, Muralidhar S A, et al.
The oxygen rich postnatal environment induces cardiomyocyte cell cycle arrest through DNA damage response.
Cell
157, 565-579 (2014)
doi: 10.1016/j.cell.2014.03.032
Members
Wataru Kimura
Team Director
Nobutoshi Ota
Visiting Scientist
Chihiro Nishiyama
Technical Staff I
Tai Sada
Junior Research Associate
Masaya Gessho
Temporary Staff
Mingxuan Ju
Student Trainee
News

Sep. 15, 2023 BDR News
Public Events
Hands-on life science workshop for high school students
Dec. 23, 2022 Research
Opossum hearts hint at new ways of fighting cardiovascular disease
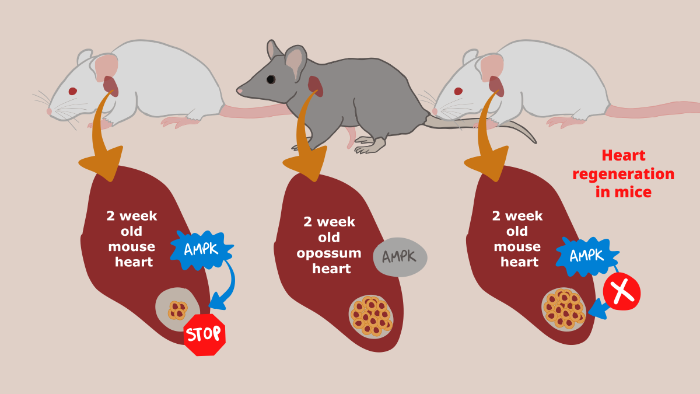
Aug. 25, 2022 Research
Harnessing the heart regeneration ability of marsupials

Jan. 21, 2022 BDR News
Dive into BDR's intriguing research
Follow the heart

Jul. 30, 2021 BDR News
RIKEN BDR signs MOU with Shinshu University for research and educational collaborations

Apr. 1, 2020 BDR News
Researcher Spotlight
Turning points and balance in a scientific career




